The Paclitaxel Paradox: Physicians Hold Steady Despite SWEDEPAD Setbacks
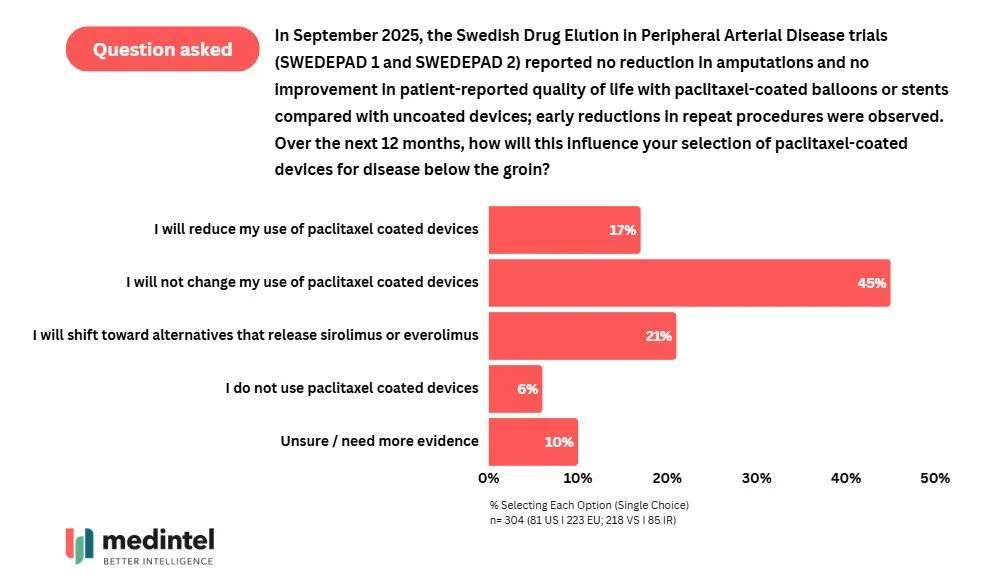
Despite trials failing to support the impact of paclitaxel-coated balloons and stents on key peripheral arterial disease endpoints, almost half of physicians do not intend to change their use patterns for paclitaxel-coated devices.
In a medintel survey of 304 physicians, 45% of vascular surgeons and interventional radiologists stated that they did not intend to change their paclitaxel-coated device practice. While 21% did indicate they would shift to sirolimus or everolimus devices, the majority seem unmoved by recent evidence.
This comes in the face of two key trials, SWEDEPAD 1 and SWEDEPAD 2, which demonstrated that paclitaxel coated devices were not associated with reduced risk of amputation or improved quality of life compared to uncoated devices in patients with chronic limb-threatening ischemia and intermittent claudication. Taken together, these findings led to principle investigators to recommend careful risk-benefit analysis when considering such devices.
The majority of physicians do not expect their paclitaxel device usage to change, despite recent clinical trial data . Surveyed in Q4 2025.
Paclitaxel technologies are no stranger to controversy in the PAD space, heralded for reducing rates of restenosis and target lesion revascularization one minute and the next, implicated in increasing mortality. The ups and downs this technology has faced in terms of clinical evidence may explain physician hesitancy to reject it on the basis of the most recent findings.
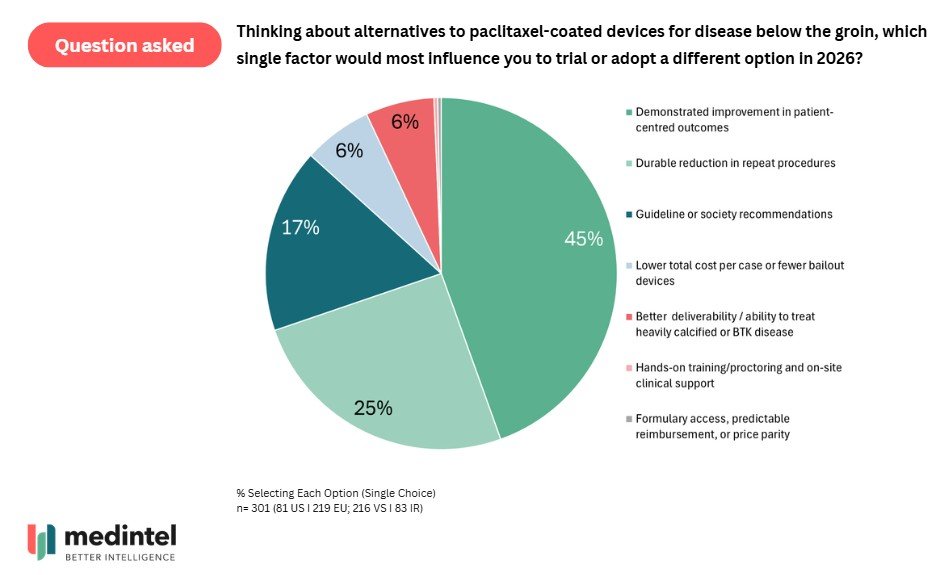
More critically, physicians appear to be waiting for a clearly superior alternative before moving away from their workhorse paclitaxel. A further line of inquiry by medintel revealed that the most influential factor driving new device utilization was demonstrated improved patient-centered outcomes, followed by a reduction in repeat procedures. This suggests that the bar for displacement isn’t reliant on cost, improved pricing structures or better training, but rather demonstratable superiority.
When considering an alternative to paclitaxel, physicians want improved patient-center outcomes and durable reductions in repeat procedures. Surveyed in Q4 2025.
This physician reluctance creates a complex market dynamic where negative evidence doesn’t immediately translate into market erosion, but where the door remains open for drug-coated technologies that can deliver on patency.
ENDS.
About the Research
Methodology Note: This analysis is based on a survey of 304 (US and EU interventional radiologists, vascular surgeons) conducted in October 2025. This survey represents a focused snapshot of perspectives rather than a comprehensive market study.
medintel is a specialist medical market research consultancy. For more information, contact intel@medintel.co.uk.
medintel conducts larger-scale quantitative and qualitative research across broader physician populations. If you're interested in more extensive PAD market research or other custom studies, please get in touch.